THE Government Analyst Food and Drug Department (GA-FDD) unearthed a number of substandard and falsified (SF) medications at nine (9) pharmacies in the city of Georgetown during a recent operation. The Guyana Police Force has been called in to assist with the investigation.
In a statement on Tuesday, GA-FDD said it launched an investigation on August 22 after receiving a complaint that an unknown individual was either clandestinely manufacturing and/ or importing SF medication for sale into the locate market. According to GA-FDD, its inspectors visited 16 pharmacies in Georgetown and outlying areas during the period August 22 to 23, 2019 and nine of the pharmacies were engaging in the sale of SF medication.

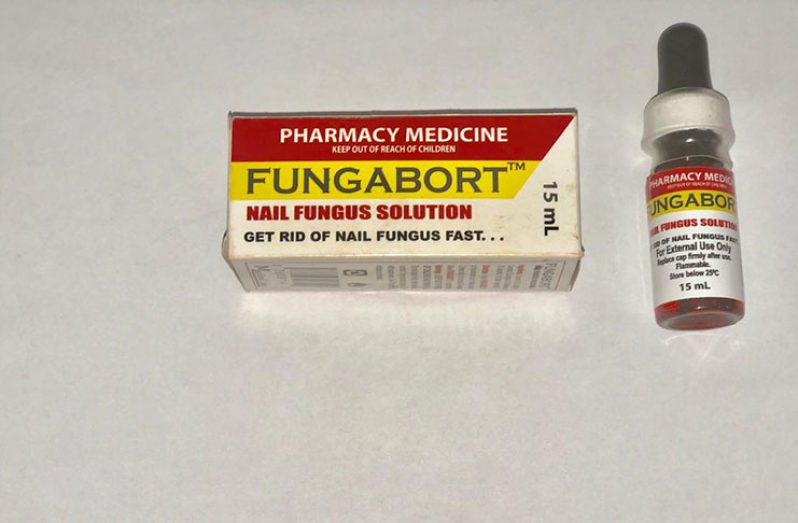
The Food and Drug watchdog said inspectors unearthed a total of 17 boxes of a medication labelled ‘chanca piedra phosmovite’ with a manufacturer’s address stated only as Mainland Labs in Canada. That medication according to the label reportedly treats gall and kidney stones, clean the liver and the urinary tract. Twenty-eight boxes of a product named ‘Fungabort’ were also found with the same manufacturer named. That ‘medication’ claims to be effective in the treatment of nail fungus. In addition, 23 boxes of Phosferine were also seized and removed from premises, since the stated address “Phosferine Health Care Co., Toronto Canada” could not have been established by the Food and Drug Department.
GA-FDD said efforts to verify the stated address on the labels of the other two products were also unsuccessful. It said that all the items were seized and removed from the premises.
“In an effort to locate the wholesaler / distributor, none of the pharmacies visited was able to furnish receipts or other documentation as proof of purchase to Inspectors. This is a clear breach of the Consumer Affairs Act Section 18: 1-3. It is also a breach of the Food and Drug’s Act, Part VI, chapter 21 section (c & d) which speaks to adequate record keeping for traceability purposes particularly for the sale of medication for patient use,” the Food and Drug Department said.
It was noted that the average retail cost for the chanca piedra phosmovite, Fungabort and Phosferine were found to be G$3500, G$ 1000 and G$3000 respectively.
“The Department is advising all pharmacies to, with immediately effect, desist from the practice of purchasing and retailing medication to be used by patients from walk-in-salesmen (suitcase traders) who refuse to provide invoices or adequate receipt or other records for traceability purposes. Inspectors will continue to seize and detain items with inaccurate addresses and where the identity of the wholesaler / distributor is unknown,” GA-FDD urged.
The Food and Drug Department has given the Guyana Police Force a detailed report to assist with the investigation. It is hoped that those responsible will be arrested.
“This we believe will ultimately ensure that, only safe and efficacious medications are released for sale on our local market,” it said.

The World Health Organization (WHO) reported in November, 2017 that SF medication distributed in low and middle income countries accounts for 10 per cent or 1 in 10 of all medication distributed, and cost these countries in excess of USD 30 million annually. The cost to the local economy and the percentage of SF medication are currently unknown.
“The GA-FDD has been able to embark upon these activities due to increased resources and personnel (Inspectors) facilitated by the Ministry of Public Health (MoPH),” it said, adding that it is currently in a substantial position to aggressively implement measures to protect consumers from the associated dangers of SF medications.
Details of the department’s exercise will be shared with members (Inspectors) at its upcoming Food Safety and Control Committee Meeting, who will be tasked with the responsibility of ensuring that pharmacies in other regions outside of Georgetown are free of the identified SF medications.





.jpg)